
Classically, kinases, which phosphorylate proteins, are regarded as the ‘on’ switches in cell signaling, while phosphatases, which dephosphorylate proteins, are thought of as the ‘off’ switches. This dynamic balance of phosphorylation and dephosphorylation is crucial in signaling pathways like the Akt pathway. Growth factors and insulin trigger PI3K activation, leading to Akt phosphorylation, which promotes cell growth and proliferation. Since excessive activation of Akt or PI3K can drive uncontrolled cell growth and proliferation (cancer), precise regulation is essential. PH domain leucine-rich repeat-containing protein phosphatases (PHLPP) have been proposed to act as negative regulators of Akt and, therefore, as tumor suppressors. However, a new study from the Leonard lab which reveals that PHLPP has no catalytic activity at all, challenges this view.
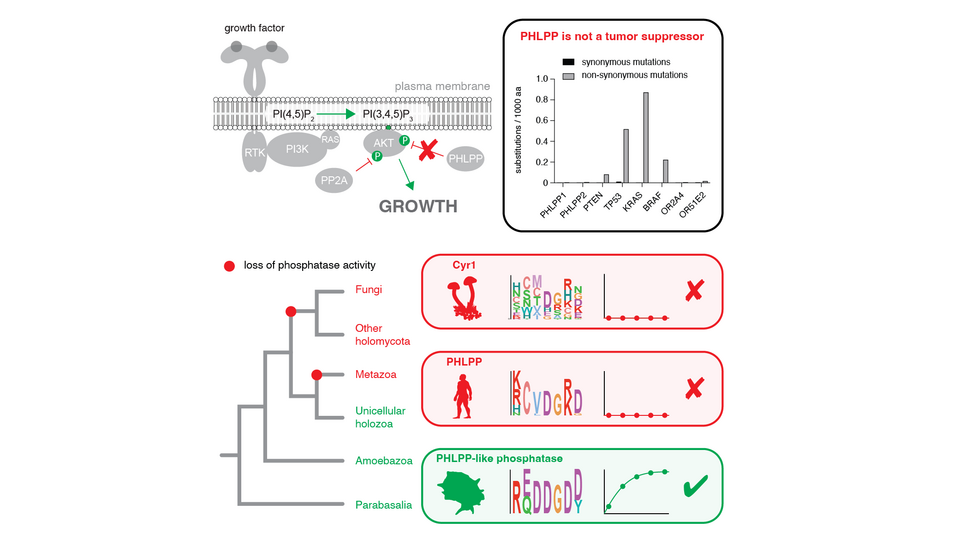
Phosphatase assays revealed that PHLPP2 lacks detectable phosphatase activity. The authors were able to trace previous reports of phosphatase activity to contamination with a different phosphatase, PP2A. Furthermore, as group leader Thomas Leonard explains: “Our structural analysis reveals that these proteins are incapable of catalyzing the phosphatase reaction.” The authors’ findings therefore prompted them to challenge the designation of PHLPP1 and PHLPP2 as tumor suppressors. The team analyzed whether somatic mutations in PHLPP1 and PHLPP2 are linked to cancer. “Unsurprisingly, our cancer genomics analysis found no evidence connecting PHLPP to cancer, raising a fundamental question: if they are neither phosphatases nor tumor suppressors, what is their true biological function? This is where the evolutionary record provides invaluable insights”, Thomas adds.
Phylogenetic analyses were conducted to trace the evolutionary history of PHLPP. The researchers discovered that PHLPP’s ancestor was a bona fide phosphatase. Around 1,000 million years ago, the ancestral gene acquired a kinase and a PH domain, leading to the formation of PHLPP. Later, in the metazoan lineage, the kinase domain was lost and the phosphatase domain was inactivated, rendering PHLPP a pseudophosphatase. Thomas elaborates: “During the course of evolution, the ancestral gene diversified into three major lineages with distinct functions. While some retained phosphatase activity, others lost it while also gaining alternative catalytic activities.” A highly conserved sub-domain of the phosphatase domain suggests the preservation of an ancient role in substrate binding and a non-catalytic, membrane scaffolding role for PHLPP in metazoans. The Leonard lab will now begin the search for the real biological function of both the ancestral phosphatase and modern-day PHLPP.
The multi-disciplinary work was supported by collaborations with experts from various fields: the Irwin lab (Gregor Mendel Institute, phylogenetics), the Haselbach lab (Institute of Molecular Pathology, cryoEM), the Bock lab (Center for Molecular Medicine, cancer genomics), as well as the Perutz MS facility and the Leeb, Ogris, and Žagrović labs.
DOI: pnas.2417218122