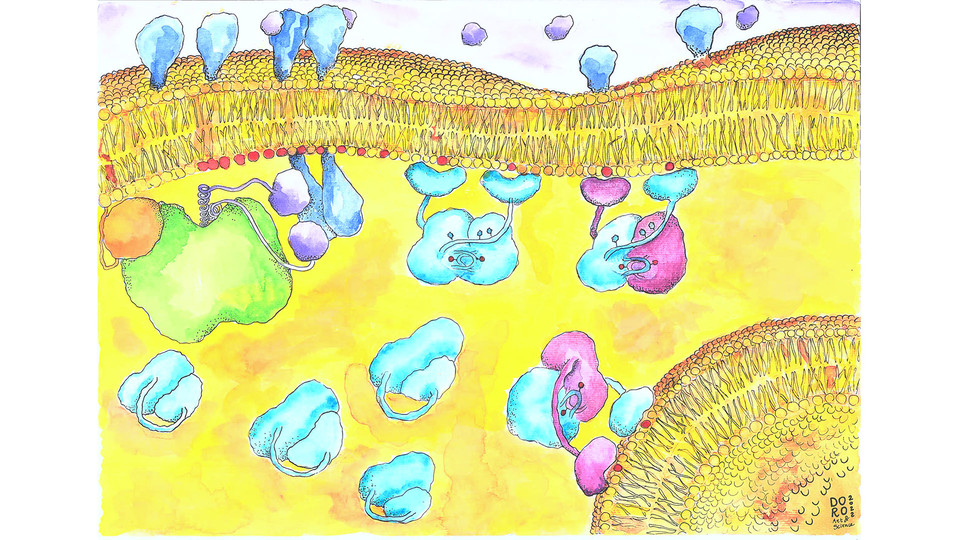

Each cell in a multicellular organism is exposed to hundreds of externals cues that are selectively transduced into appropriate cellular responses, allowing the organism to adapt to its changing environment. Key transducers of these signals are protein kinases, enzymes that propagate signals by phosphorylating downstream targets. Initiated in response to extracellular growth factors, the PI3K signaling pathway controls cell growth and proliferation and is frequently mutated in cancer. Within the PI3K pathway, the kinase PDK1 is often referred to as a ‘master kinase’, given that it regulates the activity of so many downstream signaling pathways. “In the field, it was believed that PDK1 is constitutively active, which would actually be detrimental to the cell”, says Aleksandra Levina, the lead author of the study. “If you allow the kinase to circulate freely in an active state, you not only uncouple it from the initiating signal, but you also increase the noise in the system, as it is likely to phosphorylate unwanted targets”. This apparent contradiction motivated the scientists to ask how PDK1 transduces growth factor signaling with high fidelity in a noisy cellular environment.
The scientists biochemically reconstituted PDK1 activation and show that it is acutely restricted to cellular membranes. In the absence of growth factors, they found that PDK1 exists in an autoinhibited conformation in the cell. As soon as the pathway is triggered, signaling lipids called phosphoinositides are produced in the membrane. PDK1 contains a PH domain that recognizes these lipids, specifically phosphatidylinositol-3,4,5-trisphosphate (PIP3) and phosphatidylinositol-3,4-bisphosphate PI(3,4)P2. The researchers show that PIP3 binding relieves autoinhibition of PDK1, an event that permits highly cooperative dimerization of the kinase on the membrane and switch-like activation. “Nature has evolved exquisite mechanisms to ensure that signals are propagated accurately, both in space and time”, explains group leader Thomas Leonard. “We believe that there has been a conceptual misunderstanding of what it takes to achieve this. Only by tying the activation to a membrane surface where the activating ligand is both synthesized and degraded, and not by liberation of an activated kinase, can you satisfy the requirements for both spatial and temporal control of signal propagation”.
The scientists’ findings imply that the activation of downstream kinases by PDK1, many of whom are not directly regulated by phosphoinositides, may also be dependent on PIP3 or PI(3,4)P2. With the membrane also now in focus, exciting questions about its role in these processes emerge, the scientists agree. “It is fairly intuitive to expect that if you change the surface chemistry, fluidity or lipid saturation of the membrane, you will also affect recognition of embedded lipid messengers. It will be interesting to understand precisely how the membrane composition contributes to dimerization and, ultimately, activation of PDK1”, concludes Thomas Leonard.
Aleksandra Levina, Kaelin D. Fleming, John E. Burke and Thomas A. Leonard: Activation of the essential kinase PDK1 by phosphoinositide-driven trans- autophosphorylation. Nature Communications 2022
https://doi.org/10.1038/s41467-022-29368-4